Background
Sodium Valproate (Epilim) is a medicine licensed in Ireland to treat epilepsy and bipolar disorder. If a woman becomes pregnant while taking Epilim, their baby is at risk of serious birth defects and developmental disorders. In 30-40% of cases where the child has been exposed to valproate in utero, the child will have serious developmental disorders. In 10% of cases, the child will have congenital malformations. The HSE estimate that up to 1,250 families have been impacted in these ways by exposure to valproate in pregnancy since the 1970s.
Preventative Measures
In February 2018, the European Medicines Agency (EMA) put in place measures to reduce the risk of exposure of babies to Epilim in the womb.
These measures are implemented in Ireland by the medicines regulator, the HPRA, and include:
- For women with epilepsy, Epilim must not be used in pregnancy unless there is no suitable alternative treatment.
- In female patients from the time they become able to have children, Epilim must not be used unless the conditions of a Pregnancy Prevention Programme are met.
What are the main points of the Pregnancy Prevention Programme?
- Prescribers must assess female patients for the potential of becoming pregnant, and involve the patient in evaluating her individual circumstances and supporting informed decision making,
- Pregnancy tests must be carried out before starting and during treatment as needed,
- Female patients must be informed about the risks of Epilim and what to do if planning a pregnancy or becomes pregnant
- Effective contraception must be used throughout treatment and counselling on contraception provided
- All female patients must be given the new patient information booklet and must sign an annual Risk Acknowledgement form acknowledging that they understand the risks and necessary precautions
- Importantly, all female patients must have an annual review of treatment by a specialist. If you are not currently attending specialist services, please contact your GP for advice and guidance in order to schedule a specialist review.
PLEASE CONTACT YOUR HEALTHCARE TEAM IF YOU HAVE ANY CONCERNS. DO NOT STOP TAKING VALPROATE WITHOUT FIRST SPEAKING WITH YOUR PRESCRIBER.
Epilim Patient Information booklet (June 2025)
The latest patient information booklet for Sodium Valproate can be downloaded on our website and can be accessed at the end of this page.
Resources for Healthcare Professionals
A range of resources are also available from the HPRA website for prescribers and pharmacists. These resources include a HCP Guide; posters, patient information cards and shelf-markers for pharmacists and the risk acknowledgement form for prescribers. The guide for Healthcare professionals can also be downloaded on our website or can be accessed at the end of this page.
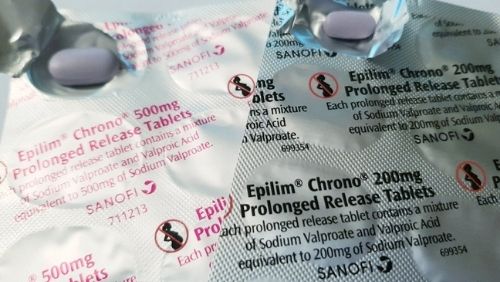
Other measures introduced in Ireland to reduce the risks associated with Epilim
- Warnings on the outside of boxes including a pictogram.
- Updated Package Insert Leaflet - these leaflets for all formulations of Epilim can be found by searching Epilim on the HPRA site
- Pack sizes have been made smaller to stop the practice of dispensing Epilim in plastic bags. The medication should be made available in its original packaging with the package insert leaflet included.
- Patient information cards should be attached to these boxes.
- Pharmacists also must follow further clear guidelines from the PSI (Pharmacy Regulator) regarding the dispensing of valproate.
Further Information
- HPRA
- HSE Valproate page
- European Medicines Agency
- Contact your local Epilepsy Ireland office
- If you took Epilim during a previous pregnancy and are concerned about your child, please get in touch with OACS Ireland (Organisation for AntiConvulsant Syndrome) or visit their Facebook Group or public Facebook page
- Contact your prescriber or your Hospital Epilepsy Helpline service
- If you are not currently attending specialist services, please contact your GP for advice and guidance in order to schedule a specialist review.
