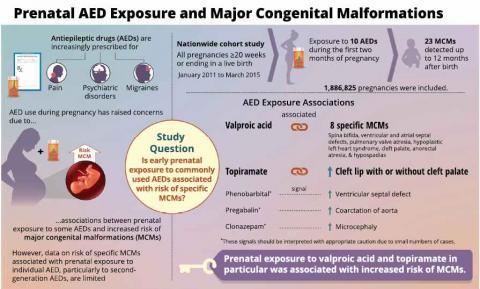
Researchers from the French National Health Insurance (CNAM) have just published a nationwide study of pregnancies ending between January 2011 and March 2015 to examine links between exposure to monotherapy with 10 different AEDs during the first two months of pregnancy and the risk of 23 specific major congenital malformations (MCMs). The study is published in the journal Neurology.
From almost 1.9m total pregnancies, the researchers found that a total 8,794 women had been prescribed an AED. 2,997 were exposed to lamotrigine (Lamictal); 1,671 to pregabalin (lyrica); 980 to clonazepam; 913 to sodium valproate (Epilim); 579 to levetiracetam (Keppra); 517 to topiramate (Topamax); 512 to carbamazepine (Tegretol); 365 to gabapentin (Neurontin); 139 to oxcarbazepine (Trileptal); and 80 to phenobarbital.
MCMs were detected up to 12 or in some cases, 24 months after birth.
Key findings included:
- Women prescribed valproate had a 19 times greater risk of having a baby with spina bifida compared with women who did not take any epilepsy drug. Valproate also increased the risk of seven other birth defects, including cleft palate and four types of heart defects.
- Women prescribed topiramate had a seven times greater risk of having a baby with cleft lip (with or without cleft palate).
- Weak signals were identified linking clonazepam to a higher risk of microcephaly (a small head circumference associated with lower IQ), while phenobarbital and pregabalin were linked to higher risks of different types of heart defects. However, the researchers have urged interpreting this data with caution because of the small numbers of cases involved.
- No significant associations in this study were found for lamotrigine, levetiracetam, carbamazepine, oxcarbazepine, or gabapentin. [Note: previous studies have associated some of these drugs with foetal malformations, for example see here]
For more on epilepsy medications in pregnancy, see our Women with Epilepsy section or download the information booklet
